Getting started
Learning a new program and/or genomic analysis is rarely as simple as one would like it to be. It might therefore be good to start the journey with one or two ‘wingmen’, so you can learn from each other. We are also arranging and participating in ChIP-seq analysis workshops – mail us if you are interested.
Installation
The download and installation proces is covered here and in this video .
Introductory book chapter
To help people getting started with basic workflows in EaSeq, we have written a chapter termed “User‐friendly and interactive analysis of ChIP‐seq data using EaSeq” for the second edition of the book Methods in Molecular Biology, Stem Cell Transcriptional Networks. We were permitted to post a copy of the chapter – Please feel free to use and share it.
Integrated assistance
We integrated some means to help you start using EaSeq within the program:
- The Demo will download a session of data (it might take a couple of minutes) that you can play around with while learning about the main features.
- The Wizard will simplify a set of basic visualization tasks.
- The Tutorials will guide you through different types of analysis and visualization and allow you to apply them on your own data.
- Finally, Hints will pop up when you start a tool or move the mouse over certain locations. They will explain the overall functionality of the feature.
input
Import mapped ChIP-seq or other -seq data as a ‘Dataset’ and regions that you would like to visualize (e.g. enhancers, CpG-islands, etc) as a ‘Regionset’. The third type of data, ‘Genesets’, contains gene annotation information and is typically downloaded directly within EaSeq. You can read more about the input types and how to obtain data here as well as watch a video on how to import data here.
For newcommers
Upcoming workshops can be found here.
We also made a collection of videos for new users who prefer watching how to do things first. If you would like to learn a particular workflow that is not covered or have a figure from a paper that you would like to reproduce, then we will gladly consider it for a new video – so please mail us.
A list of the current videos:
- How to download and install EaSeq.
- Introducing the main interface.
- How to import ChIP-seq data and a set of genomic regions into EaSeq (the example data can be downloaded here).
- How to make your first heatmaps from the imported data above. In the example we visualize histone marks (H3K27me3 and H3K36me3) at CpG-islands and sort them according to their sizes.
- How to access the autogenerated legends and methods.
- How to make superimposed tracks. In the example we visualize histone marks (H3K27me3 and H3K36me3) at CpG-islands.
- How to export plots as tiff files and toggle autoenumeration on/off.
- How to use EaSeq as a genome browser and explore the underlying loci in a heatmap.
- How to download gene annotation data and extract TSSes as new regions.
- How to gate out regions and instruct new plots to show this population.
- How to handle plots and change their settings.
- EaSeq also contains a set of integrated tutorials that can guide you through how to apply a particular visualization or analysis to your own data. Learn how to run these as well as the the integrated demo from within EaSeq.
(All of the steps above are also combined in this 27-minute video)
- How to make heatmaps of ChIP-seq signal at genes and sort them according to sizes.
- How to make ratiometric heatmaps, calculate fold difference, and sort the regions accordingly.
Getting help
EaSeq also contains an integrated chat forum where you can send Squeaks.
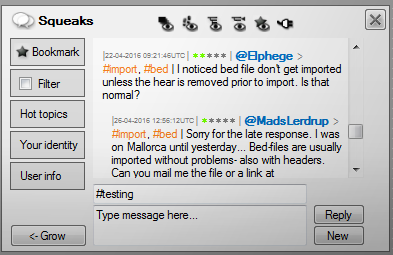
If you post a Squeak containing a question, then it will be broadcasted to all online EaSeq users immediately. We encourage new users to get help this way, and more experienced users to help others and post replies.
For general questions on sequencing data analysis, then we would also strongly recommend using the websites Biostars and seqanswers. They are full of already asked questions from newbies and experts as well as answers from experienced bioinformaticians.
Finally, we have a section for Frequently Asked Questions, and if you can’t find an answer for your question then please feel free to mail us.